This file is licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license. GFDL GNU Free Documentation License true true 
A copy of the license is included in the section entitled GNU Free Documentation License. Permission is granted to copy, distribute and/or modify this document under the terms of the GNU Free Documentation License, Version 1.2 or any later version published by the Free Software Foundation with no Invariant Sections, no Front-Cover Texts, and no Back-Cover Texts. The study was supported by the NEI Intramural Research Program, grants ZIAEY000450 and ZIAEY000546.I, the copyright holder of this work, hereby publish it under the following licenses: Reference: “High-resolution genome topology of human retina uncovers super enhancer-promoter interactions at tissue-specific and multifactorial disease loci” by Claire Marchal, Nivedita Singh, Zachary Batz, Jayshree Advani, Catherine Jaeger, Ximena Corso-Díaz and Anand Swaroop, 7 October 2022, Nature Communications. 
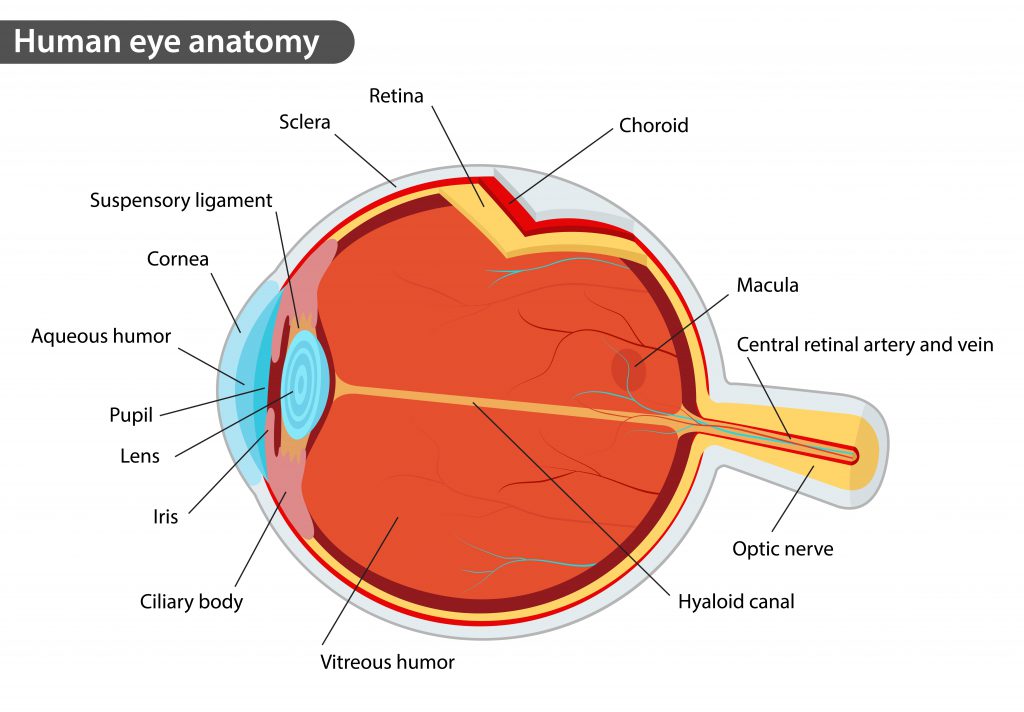
The integrated genome regulatory map will also assist in evaluating genes associated with other common retina-associated diseases such as diabetic retinopathy, determining missing heritability, and understanding genotype-phenotype correlations in inherited retinal and macular diseases. The findings point to specific candidate causal genes involved in those diseases. The scientists integrated the chromatin topology map with data on genetic variants identified from genome-wide association studies for their involvement in age-related macular degeneration (AMD) and glaucoma, two of the leading causes of vision loss and blindness. More than a third (35.7%) of gene pairs interacting through a chromatin loop in mice also did so in human retina. Moreover, similarities between mice and human chromatin organization suggest conservation across species, underscoring the relevance of chromatin organizational patterns for retinal gene regulation. “Having such a high-resolution picture of genomic architecture will continue to provide insights into the genetic control of tissue-specific functions,” Swaroop said. They found distinct patterns of interaction at retinal genes suggesting how chromatin’s 3D organization plays an important role in tissue-specific gene regulation. What emerged was a dynamic picture of interactions within chromatin over time, including gene activity hot spots and areas with varying degrees of insulation from other regions of DNA. The research team then integrated that chromatin topology map with datasets on retinal genes and regulatory elements. Maps were constructed using post-mortem retinal samples from four human donors. Using deep Hi-C sequencing, a tool used for studying 3D genome organization, the scientists created a high-resolution map that included 704 million contact points within retinal cell chromatin. This has shed light on the specific mechanisms by which non-coding regulatory elements exert control even when their location on a DNA strand is remote from the genes they regulate. Many of the proteins - namely, histones - package the massive amount of DNA in a genome into a highly compact form that can fit in the cell nucleus.įor a long time, such non-coding sequences were considered “junk DNA.” However, more advanced studies have demonstrated ways that these sequences control which genes get transcribed and when. He is senior investigator and chief of the Neurobiology Neurodegeneration and Repair Laboratory at the NEI, part of the National Institutes of Health (NIH).Ĭhromatin is a mixture of DNA and proteins that form the chromosomes found in the cells of humans and other higher organisms. “This is the first detailed integration of retinal regulatory genome topology with genetic variants associated with age-related macular degeneration (AMD) and glaucoma, two leading causes of vision loss and blindness,” said Anand Swaroop, Ph.D., the study’s lead investigator. The study will be published today (October 7, 2022) in the journal Nature Communications. The resulting comprehensive gene regulatory network provides insights into the regulation of gene expression in general, and in retinal function, in both rare and common eye diseases. These are the fibers that package 3 billion nucleotide-long DNA molecules into compact structures that fit into chromosomes within each cell’s nucleus. National Eye Institute (NEI) scientists have mapped the organization of human retinal cell chromatin. NIH researchers reveal new insights on how genetic architecture determines gene expression, tissue-specific function, and disease phenotype in blinding diseases. Researchers mapped the organization of human retinal cell chromatin, resulting in a comprehensive gene regulatory network that provides insights into the regulation of gene expression in both rare and common eye diseases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
